GREEN CHEMICAL PRODUCTION BASED ON THERMAL CRACKING OF INEDIBLE VEGETABLE OIL
Original scientific paper
DOI:
https://doi.org/10.2298/CICEQ220114033CKeywords:
Triglyceride pyrolysis, castor oil, ricinoleic acid, heptaldehyde, undecylenic acid, methyl undecenoateAbstract
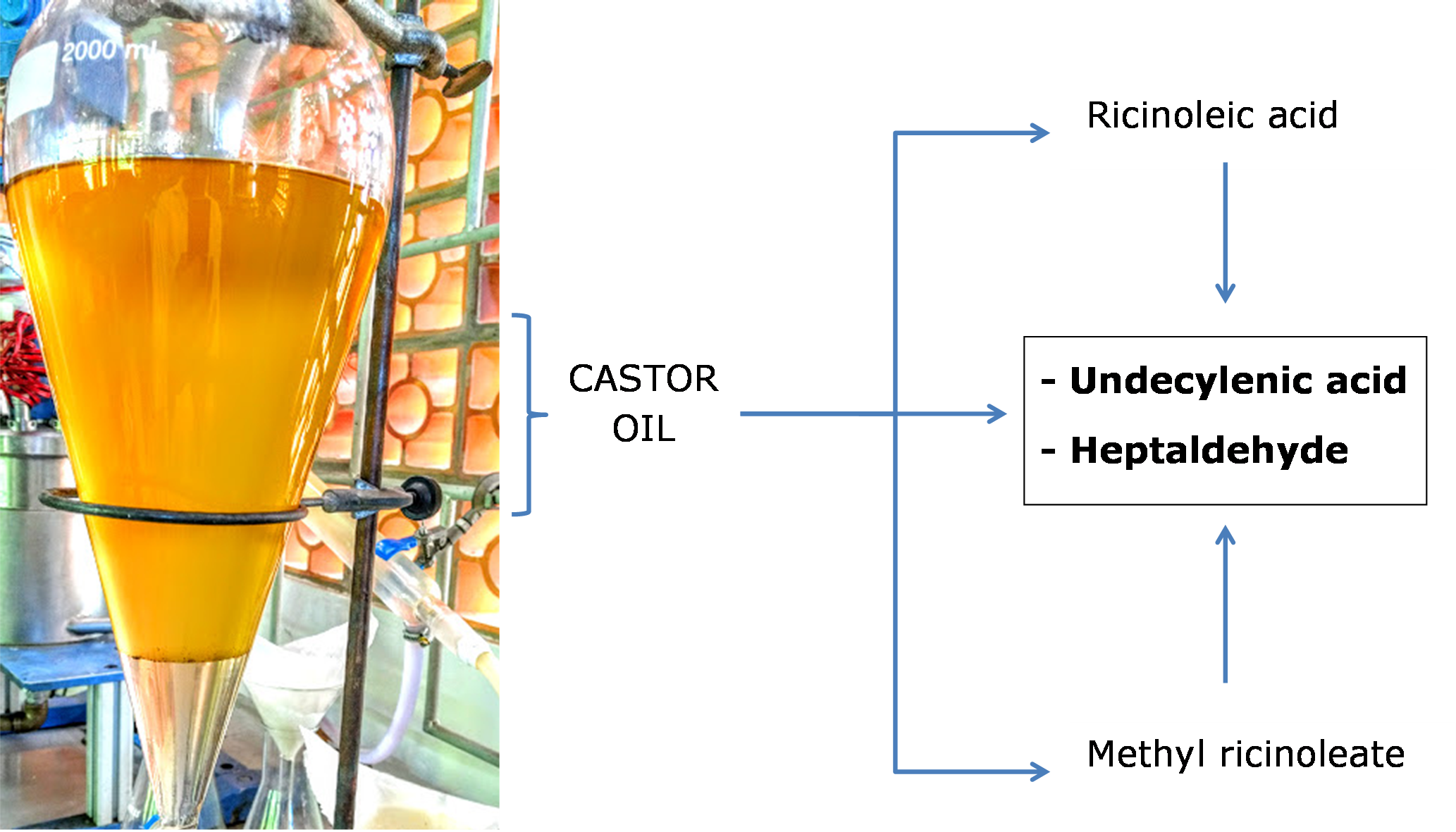
This work evaluated the process for heptaldehyde, undecylenic acid, and methyl undecenoate production from castor oil, methyl ester of castor oil, and ricinoleic acid. Experiments were performed in a continuous pilot-plant scale pyrolysis reactor. Those are very important green chemical products that the thermal cracking of castor oil might produce. Transesterification of castor oil produces methyl ricinoleate, and its thermal cracking generates methyl undecenoate and heptaldehyde. The pyrolysis temperatures tested were 530 °C, 545 °C, 560 °C, and 575 °C, with residence time from 17 s to 32 s and mass flow at 400 g/h of the mixture of materials with 25% distilled water. It was observed that the temperature influenced the bio-oil yield in different degree for each material. The bio-oil was characterized by iodine index, acid number, and mass, and the contents of its compounds were obtained by GC-FID chromatography. The best result for the undecylenic acid mass yield of the desired compounds occurred at 530 °C, achieving 17.8% from ricinoleic acid and 16.5% from castor oil. For the heptaldehyde, the highest production was also obtained at 530 °C, with a value of 20.7% from methyl ester and 15.2% from ricinoleic acid.
References
H. Mutlu, M.A.R. MeierEur. J. Lipid Sci. Technol. 112 (2010) 10—30. http://dx.doi.org/10.1002/ejlt.200900138.
X. Mao, Q. Xie, Y. Duan, S. Yu, Y. Nie, Appl. Therm. Eng. 194 (2021) 117093. http://dx.doi.org/10.3390/ma15041565.
E.B. Mubofu, Sustain. Chem. Process. 4 (2016). https://doi.org/10.1186/s40508-016-0055-8.
M. Van der Steen, C.V. Stevens, ChemSusChem. 2, (2009) 692—713. https://doi.org/10.1002/cssc.200900075.
W.J. Kh'ng, W.A. Ibrahim, Z. Hassan, I. Izirwan, Energy Reports. 8, (2022) 11—18. https://doi.org/10.1016/j.egyr.2022.10.111.
S. Singh, S. Sharma, S.J Sarma, S.K. Brar, Environ. Prog. Sustain. Energy. 42 (2022) E14008. https://doi.org/10.1002/ep.14008.
FAOSTAT Statistical Database (2019), Food and Agriculture Organization of the United Nations, http://www.fao.org/faostat/en/#compare [accessed 7 December 2021.
V. Botton, R. Torres De Souza, V.R. Wiggers, D.R. Scharf, E.L. Simionatto, L. Ender, H.F. Meier, J. Anal. Appl. Pyrolysis 121 (2016) 387—393. http://dx.doi.org/10.1016/j.jaap.2016.09.002.
G. Das, R.K. Trivedi, JAOCS 66 (1989) 938—941. https://doi.org/10.1007/BF02682613.
C. Tang, Z. Yuan, Catalytic cracking method for preparing 10-undecenoic acid and heptaldehyde - PCT CN103819330 (2014).
G. Wetroff, L. Thillay, G. Divachetf, J. Khaladji, Pyrolysis of Ricinoleates - PCT US2807633 (1957).
H. Guobin, L. Zuyu, Y. Suling, Y. Rufeng, JAOCS 73 (1996) 1109—1112. https://doi.org/10.1007/BF02523370.
G. Menshhein, V. Costa, L.M. Chiarello, D.R. Scharf, E.L. Simionato, V. Botton, H.F. Meier, V.R. Wiggers, L. Ender, Renew. Energy 142 (2019) 561—568. https://doi.org/10.1016/j.renene.2019.04.136.
G. Menshhein, V. Costa, L.M. Chiarello, D.R. Scharf, E.L. Simionato, V. Botton, H.F. Meier, V.R. Wiggers, L. Ender, Data Br. 25 (2019) 104325. https://doi.org/10.1016/j.dib.2019.104325.
K. Ramezani, S. Rowshanzamir, M.H. Eikani, Energy 35 (2010) 4142—4148. https://doi.org/10.1016/j.energy.2010.06.034.
V. Botton, D.R. Riva, E.L. Simionatto, V.R. Wiggers, L. Ender, H.F. Meier, A.A.C. Barros, Quim. Nova 35 (2012) 677—682. https://doi.org/10.1590/S0100-40422012000400004.
H.F. Meier, V.R. Wiggers, G.R. Zonta, D.R. Scharf, E.L. Simionatto, L. Ender, Fuel 144 (2015) 50—59. https://doi.org/10.1016/j.fuel.2014.12.020.
R.F. Beims, V. Botton, L. Ender, D.R. Scharf, E.L. Simionatto, H.F. Meier, V.R. Wiggers, Fuel 217 (2018) 175—184. https://doi.org/10.1016/j.fuel.2017.12.109.
D.P. Matharasi, G. Ramya, A. Asha, P. Jayaprakash. J. Indian Chem. Soc. 99, (2022) 100757. https://doi.org/10.1016/j.jics.2022.100757.
R. Ganesan, S. Subramaniam, R. Paramasivam, J.S.M. Sabir, J.S. Femilda Josephin, K. Brindhadevi, A. Pugazhendhi, Sci. Total Environ. 757 (2021) 143781. https://doi.org/10.1016/j.scitotenv.2020.143781.
C.A. Canciam, E-xacta 4 (2011) 7—18. http://dx.doi.org/10.18674/exacta.v4i3.667.
I.G. Prabasari, R. Sarip, S. Rahmayani, N. Nazarudin, Makara J. Sci. 23 (2019) 162—168. https://doi.org/10.7454/mss.v23i3.11264.
N. Nazarudin, U. Ulyarti, O. Alfernando, Y.Y. Hans, Reaktor. 22, (2022) 21—27. https://doi.org/10.14710/reaktor.22.1.21-27.
N. Nazarudin, I.G. Prabasari, U. Ulyarti, Susilawati, A. Oktadio, J. Phys. Conf. Ser. 1567 (2020) 022021. https://doi.org/10.1088/1742-6596/1567/2/022021.
Downloads
Published
Issue
Section
License
Copyright (c) 2021 Vanderlei Rodrigues Costa, Luana Marcele Chiarello, Vanderleia Botton, Edésio Luiz Simionatto , Vinicyus Rodolfo Wiggers, Henry França Meier , Laércio Ender

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish with this journal agree to the following terms:
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Authors grant to the Publisher the following rights to the manuscript, including any supplemental material, and any parts, extracts or elements thereof:
- the right to reproduce and distribute the Manuscript in printed form, including print-on-demand;
- the right to produce prepublications, reprints, and special editions of the Manuscript;
- the right to translate the Manuscript into other languages;
- the right to reproduce the Manuscript using photomechanical or similar means including, but not limited to photocopy, and the right to distribute these reproductions;
- the right to reproduce and distribute the Manuscript electronically or optically on any and all data carriers or storage media – especially in machine readable/digitalized form on data carriers such as hard drive, CD-Rom, DVD, Blu-ray Disc (BD), Mini-Disk, data tape – and the right to reproduce and distribute the Article via these data carriers;
- the right to store the Manuscript in databases, including online databases, and the right of transmission of the Manuscript in all technical systems and modes;
- the right to make the Manuscript available to the public or to closed user groups on individual demand, for use on monitors or other readers (including e-books), and in printable form for the user, either via the internet, other online services, or via internal or external networks.
How to Cite
Funding data
-
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior
Grant numbers 001