Procena efikasnosti inovativnog reagensa za prečišćavanje vode iz reke Save (Beograd) Naučni rad
Glavni sadržaj članka
Apstrakt
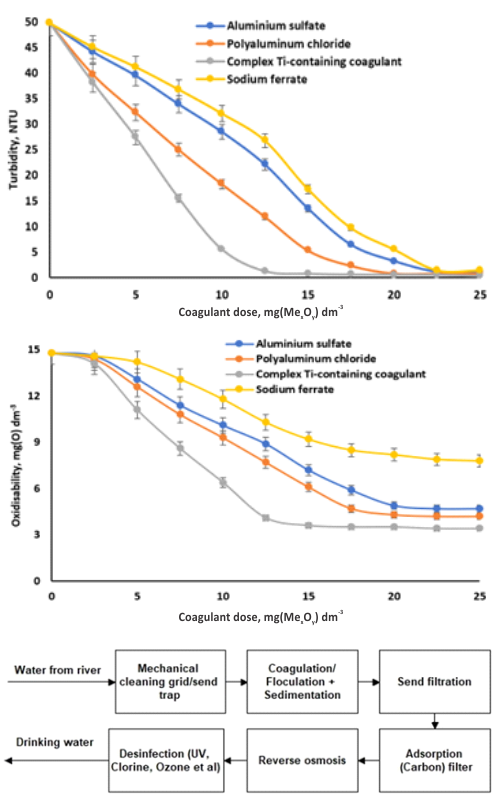
U Srbiji se sve više pažnje posvećuje unapređenju ekološke bezbednosti. Razvoj tehnologija za zaštitu hidrosfere predstavlja jedan od prioritetnih zadataka, čije će rešavanje značajno poboljšati kvalitet života stanovništva i približiti zemlju standardima Evropske unije. Reka Sava je jedan od najvećih vodotokova koji protiču kroz Beograd i predstavlja važan resurs koji zahteva efikasno upravljanje kvalitetom vode. U ovom radu je pokazano da primena kompleksnih koagulanata koji sadrže titanijum omogućava ne samo efikasno uklanjanje dispergovanih čestica iz vode i smanjenje sadržaja organskih jedinjenja za 70 %, već i smanjenje nivoa mikrobiološke kontaminacije za 66 %. Upotreba natrijum-ferata, koji deluje i kao koagulant i kao baktericid, omogućava značajno smanjenje sadržaja zagađujućih materija u vodi, kao i potpunu dekontaminaciju vode. Takođe je pokazano da kompleksni koagulacioni reagens koji sadrži titanijum značajno povećava brzinu sedimentacije koagulacionog mulja za 20 do 30 % i brzinu filtracije za 10 do 20 %.
Detalji članka
Broj časopisa
Rubrika

Ovaj rad je pod Creative Commons Autorstvo 4.0 Internacionalna licenca.
Kada je rukopis prihvaćen za objavlјivanje, autori prenose autorska prava na izdavača. U slučaju da rukopis ne bude prihvaćen za štampu u časopisu, autori zadržavaju sva prava.
Na izdavača se prenose sledeća prava na rukopis, uklјučujući i dodatne materijale, i sve delove, izvode ili elemente rukopisa:
- pravo da reprodukuje i distribuira rukopis u štampanom obliku, uklјučujući i štampanje na zahtev;
- pravo na štampanje probnih primeraka, reprint i specijalnih izdanja rukopisa;
- pravo da rukopis prevede na druge jezike;
- pravo da rukopis reprodukuje koristeći fotomehanička ili slična sredstva, uklјučujući, ali ne ograničavajući se na fotokopiranje, i pravo da distribuira ove kopije;
- pravo da rukopis reprodukuje i distribuira elektronski ili optički koristeći sve nosioce podataka ili medija za pohranjivanje, a naročito u mašinski čitlјivoj/digitalizovanoj formi na nosačima podataka kao što su hard disk, CD-ROM, DVD, Blu-ray Disc (BD), mini disk, trake sa podacima, i pravo da reprodukuje i distribuira rukopis sa tih prenosnika podataka;
- pravo da sačuva rukopis u bazama podataka, uklјučujući i onlajn baze podataka, kao i pravo prenosa rukopisa u svim tehničkim sistemima i režimima;
- pravo da rukopis učini dostupnim javnosti ili zatvorenim grupama korisnika na osnovu pojedinačnih zahteva za upotrebu na monitoru ili drugim čitačima (uklјučujući i čitače elektonskih knjiga), i u štampanoj formi za korisnike, bilo putem interneta, onlajn servisa, ili putem internih ili eksternih mreža.
Kako citirati
Reference
[1] Dettori M, Arghittu A, Deiana G, Castiglia P, Azara A. The revised European Directive 2020/2184 on the quality of water intended for human consumption. A step forward in risk assessment, consumer safety and informative communication. Environ Res. 2022; 20: 112773. https://doi.org/10.1016/j.envres.2022.112773
[2] Marković M, Zuliani T, Simić SB, Mataruga Z, Kostić O, Jarić S, Vidmar J, Milačič R, Ščančar J, Mitrović M, Pavlović P. Potentially toxic elements in the riparian soils of the Sava River. J Soil Sediments. 2018; 18: 3404-3414. https://doi.org/10.1007/s11368-018-2071-7
[3] Babić G, Vuković M, Voza D, Takić L, Mladenović-Ranisavljević I. Assessing Surface Water Quality in the Serbian Part of the Tisa River Basin. Polish J Environ Stud. 2019; 28(6); 4073-4085. https://doi.org/10.15244/pjoes/95184
[4] Kašanin-Grubin M, Gajić V, Veselinović G, Stojadinović S, Antić N, Štrbac S. Provenance and Pollution Status of river Sediments in the Danube Watershed in Serbia. Water. 2023; 15(19): 3406. https://doi.org/10.3390/w15193406
[5] Stevanović SD, Krstić J., Stojanović BT, Paunović DĐ, Dimitrijević DS, Veličković JM, Stanković NJ. Monitoring of drinking water from the karst springs of the Ljuberadja-Niš water supply system (Serbia). SN Appl Sci. 2020; 2: 1847. https://doi.org/10.1007/s42452-020-03674-2
[6] Han SW, Kang June LS. Comparison of Al(III) and Fe(III) Coagulants for Improving Coagulation Effectiveness in Water Treatment. J Korean Soc Env Eng. 2015; 37: 325-331. https://doi.org/10.4491/KSEE.2015.37.6.325
[7] Gan Y, Li J, Zhang L, Bi W, Huang W, Li H, Zhang S. Potential of titanium coagulants for water and wastewater treatment: Current status and future perspectives. Chem Eng J. 2021; 406: 126837. https://doi.org/10.1016/j.cej.2020.126837
[8] Thomas M, Bąk J, Królikowska J. Efficiency of titanium salts as alternative coagulants in water and wastewater treatment. Desalin Water Treat. 2020; 208: 261-272. 10.5004/dwt.2020.26689
[9] Kuzin EN, Krutchinina NE. Evaluation of effectiveness of use of complex coagulants for wastewater treatment processes of mechanical engineering. ChemChemTech. 2019; 62 (10): 140-146. https://doi.org/10.6060/ivkkt.20196210.5939
[10] Kuzin E. Synthesis and Use of Complex Titanium-Containing Coagulant in Water Purification Processes. Inorganics. 2025; 13 (1): 9. https://doi.org/10.3390/inorganics13010009
[11] Thomas M, Zdebik D. Treatment of Real Textile Wastewater by Using Potassium Ferrate(VI) and Fe(III)/H2O2. Application of Aliivibrio Fischeri and Brachionus plicatilis. Tests for Toxicity Assessment. Fibr and Text East Europe. 2019; 27(3): 78-84. https://doi.org/10.5604/01.3001.0013.0746
[12] Thomas M, Kozik V, Barbusiński K, Sochanik A, Jampilek J, Bak A. Potassium Ferrate (VI) as the Multifunctional Agent in the Treatment of Landfill Leachate. Materials. 2020; 13 (21): 5017. https://doi.org/10.3390/ma13215017
[13] Yu J, Zhang SK, Zhu Q, Wu C, Huang S, Zhang Y, Yao S, Pang W. A Review of Research Progress in the Preparation and Application of Ferrate (VI). Water. 2023; 15: 699. https://doi.org/10.3390/ma13215017
[14] Talaiekhozani A, Talaei MR, Rezania SN. An overview on production and application of ferrate (VI) for chemical oxidation, coagulation and disinfection of water and wastewater. J Envir Chem Eng. 2017; 5(2): 1828-1842. https://doi.org/10.1016/j.jece.2017.03.025
[15] Ghernaout D, Naceur MW. Ferrate (VI): In situ generation and water treatment. Desalin Water Treat. 2011;
30(1-3): 1-14. https://doi.org/10.5004/dwt.2011.2217
[16] Munyengabe A, Zvinowanda C. Production, characterization and application of Ferrate(VI) in water and wastewater treatments. Brazilian Journal of Analytical Chemistry. 2019; 6(25): 40-57. https://doi.org/10.30744/brjac.2179-3425.RV-19-2019
[17] Nyzhnyk T. High efficiency titanium coagulants for water treatment. TT: PhE. 2017; 1: 54-56. http://dx.doi.org/10.21303/2585-6847.2017.00486
[18] Zhao YX, Li XY. Polymerized titanium salts for municipal wastewater preliminary treatment followed by further purification via crossflow filtration for water reuse. Sep Purif Technol. 2019; 211: 207-217. https://doi.org/10.1016/j.seppur.2018.09.078
[19] Men Y, Wang X, Cheng S, Zhu L, Li Z. Pre-coagulation with novel titanium coagulants for mitigating membrane fouling in direct membrane filtration of municipal wastewater. Chem Eng J. 2024; 495: 153156. https://doi.org/10.1016/j.cej.2024.153156
[20] Gan Y, Zhang L, Zhang S. The suitability of titanium salts in coagulation removal of micropollutants and in alleviation of membrane fouling. Water Res. 2021; 205: 117692. https://doi.org/10.1016/j.watres.2021.117692
[21] Calderón AJ, González I. Some Hardware and Instrumentation Aspects of the Development of an Automation System for Jar Tests in Drinking Water Treatment. Sensors. 2017; 17: 2305. https://doi.org/10.3390/s17102305
[22] Sarantseva AA, Ivantsova NA, Kuzin EN. Investigation of the Process of Oxidative Degradation of Phenol by Sodium Ferrate Solutions. Russ J Gen Chem. 2023; 93(13): 3454-3459. https://doi.org/10.1134/S1070363223130273
[23] ISO 15705:2002. Water Quality—Determination of the Chemical Oxygen Demand Index (ST‐COD)‐SMALL‐Scale Sealed‐Tube Method; ISO: Geneva, Switzerland. 2002. https://www.iso.org/standard/28778.html
[24] Bain R, Bartram J, Elliott M, Matthews R, McMahan L, Tung R, Chuang P, Gundry S. A summary catalogue of microbial drinking water tests for low and medium resource settings. Int J Environ Res Public Health. 2012; 9: 1609-1625. https://doi.org/10.3390/ijerph9051609
[25] Collivignarelli MC, Abbà A, Benigna I, Sorlini S, Torretta V. Overview of the Main Disinfection Processes for Wastewater and Drinking Water Treatment Plants. Sustainability. 2018; 10(1): 86. https://doi.org/10.3390/su10010086
[26] Kuzin EN, Kruchinina NE. Titanium-containing coagulants. for foundry wastewater treatment. CIS Iron and Steel Review. 2020; 20(2): 66-69. https://doi.org/10.17580/cisisr.2020.02.14
[27] Wang TH, Navarrete‐López AM, Li S, Dixon DA, Gole JL. Hydrolysis of TiCl4: initial steps in the production of TiO2. J Phys Chem A. 2010; 114: 7561-7570. http://dx.doi.org/10.1021/jp102020h
[28] Chekli L, Eripret C, Park S, Tabatabai S, Vronska O, Tamburic B, Kim J, Shon HK. Coagulation performance and floc characteristics of polytitanium tetrachloride (PTC) compared with titanium tetrachloride (TiCl4) and ferric chloride (FeCl3) in algal turbid water. Sep Purif Technol. 2017; 175: 99-106. https://doi.org/10.1016/j.seppur.2016.11.019.
[29] Michael Berg J., Romoser A., Banerjee N. Zebda R., Sayes C. M. The relationship between pH and zeta potential of ∼ 30 nm metal oxide nanoparticle suspensions relevant to in vitro toxicological evaluations. Nanotoxicology. 2009; 3-4: 276-283. https://doi.org/10.3109/17435390903276941