Assessment of the efficiency of innovative reagents for purification of water from the Sava River (Belgrade) Original scientific paper
Main Article Content
Abstract
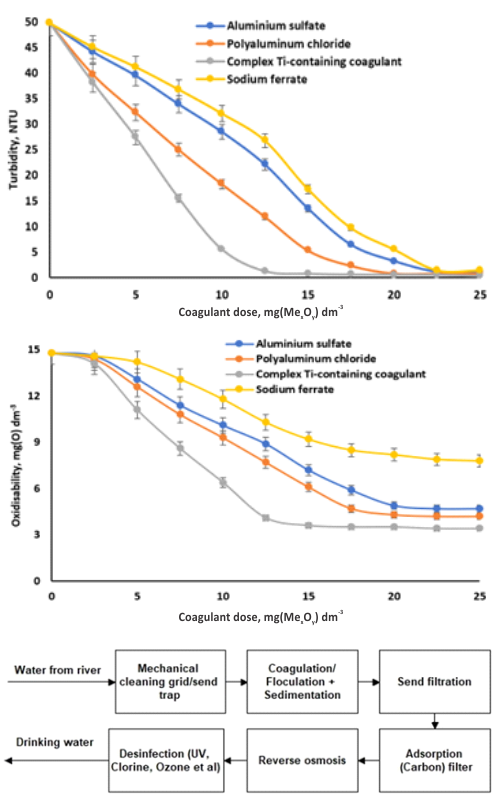
There is a growing focus on enhancing environmental safety in Serbia. The development of hydrosphere protection technologies is one of the priority tasks, the solution of which will considerably improve the quality of life for the population and bring the country closer to the EU standards. The Sava River is one of the largest waterways running through Belgrade. In this work it was shown that the use of complex titanium-containing coagulants allows not only for the efficient removal of dispersed particles from water and a 70 % reduction in organic compound content, but also a 66 % decrease in the level of microbiological contamination. The use of sodium ferrate, a coagulant and bactericide, allows for a significant reduction of pollutant content in water as well as complete water decontamination. It was demonstrated that the use of a complex titanium-containing reagent considerably increases the sedimentation rate of coagulation sludge by 20 to 30 % and the filtration rate by 10 to 20 %.
Article Details
Issue
Section

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Authors grant to the Publisher the following rights to the manuscript, including any supplemental material, and any parts, extracts or elements thereof:
- the right to reproduce and distribute the Manuscript in printed form, including print-on-demand;
- the right to produce prepublications, reprints, and special editions of the Manuscript;
- the right to translate the Manuscript into other languages;
- the right to reproduce the Manuscript using photomechanical or similar means including, but not limited to photocopy, and the right to distribute these reproductions;
- the right to reproduce and distribute the Manuscript electronically or optically on any and all data carriers or storage media – especially in machine readable/digitalized form on data carriers such as hard drive, CD-Rom, DVD, Blu-ray Disc (BD), Mini-Disk, data tape – and the right to reproduce and distribute the Article via these data carriers;
- the right to store the Manuscript in databases, including online databases, and the right of transmission of the Manuscript in all technical systems and modes;
- the right to make the Manuscript available to the public or to closed user groups on individual demand, for use on monitors or other readers (including e-books), and in printable form for the user, either via the internet, other online services, or via internal or external networks.
How to Cite
References
[1] Dettori M, Arghittu A, Deiana G, Castiglia P, Azara A. The revised European Directive 2020/2184 on the quality of water intended for human consumption. A step forward in risk assessment, consumer safety and informative communication. Environ Res. 2022; 20: 112773. https://doi.org/10.1016/j.envres.2022.112773
[2] Marković M, Zuliani T, Simić SB, Mataruga Z, Kostić O, Jarić S, Vidmar J, Milačič R, Ščančar J, Mitrović M, Pavlović P. Potentially toxic elements in the riparian soils of the Sava River. J Soil Sediments. 2018; 18: 3404-3414. https://doi.org/10.1007/s11368-018-2071-7
[3] Babić G, Vuković M, Voza D, Takić L, Mladenović-Ranisavljević I. Assessing Surface Water Quality in the Serbian Part of the Tisa River Basin. Polish J Environ Stud. 2019; 28(6); 4073-4085. https://doi.org/10.15244/pjoes/95184
[4] Kašanin-Grubin M, Gajić V, Veselinović G, Stojadinović S, Antić N, Štrbac S. Provenance and Pollution Status of river Sediments in the Danube Watershed in Serbia. Water. 2023; 15(19): 3406. https://doi.org/10.3390/w15193406
[5] Stevanović SD, Krstić J., Stojanović BT, Paunović DĐ, Dimitrijević DS, Veličković JM, Stanković NJ. Monitoring of drinking water from the karst springs of the Ljuberadja-Niš water supply system (Serbia). SN Appl Sci. 2020; 2: 1847. https://doi.org/10.1007/s42452-020-03674-2
[6] Han SW, Kang June LS. Comparison of Al(III) and Fe(III) Coagulants for Improving Coagulation Effectiveness in Water Treatment. J Korean Soc Env Eng. 2015; 37: 325-331. https://doi.org/10.4491/KSEE.2015.37.6.325
[7] Gan Y, Li J, Zhang L, Bi W, Huang W, Li H, Zhang S. Potential of titanium coagulants for water and wastewater treatment: Current status and future perspectives. Chem Eng J. 2021; 406: 126837. https://doi.org/10.1016/j.cej.2020.126837
[8] Thomas M, Bąk J, Królikowska J. Efficiency of titanium salts as alternative coagulants in water and wastewater treatment. Desalin Water Treat. 2020; 208: 261-272. 10.5004/dwt.2020.26689
[9] Kuzin EN, Krutchinina NE. Evaluation of effectiveness of use of complex coagulants for wastewater treatment processes of mechanical engineering. ChemChemTech. 2019; 62 (10): 140-146. https://doi.org/10.6060/ivkkt.20196210.5939
[10] Kuzin E. Synthesis and Use of Complex Titanium-Containing Coagulant in Water Purification Processes. Inorganics. 2025; 13 (1): 9. https://doi.org/10.3390/inorganics13010009
[11] Thomas M, Zdebik D. Treatment of Real Textile Wastewater by Using Potassium Ferrate(VI) and Fe(III)/H2O2. Application of Aliivibrio Fischeri and Brachionus plicatilis. Tests for Toxicity Assessment. Fibr and Text East Europe. 2019; 27(3): 78-84. https://doi.org/10.5604/01.3001.0013.0746
[12] Thomas M, Kozik V, Barbusiński K, Sochanik A, Jampilek J, Bak A. Potassium Ferrate (VI) as the Multifunctional Agent in the Treatment of Landfill Leachate. Materials. 2020; 13 (21): 5017. https://doi.org/10.3390/ma13215017
[13] Yu J, Zhang SK, Zhu Q, Wu C, Huang S, Zhang Y, Yao S, Pang W. A Review of Research Progress in the Preparation and Application of Ferrate (VI). Water. 2023; 15: 699. https://doi.org/10.3390/ma13215017
[14] Talaiekhozani A, Talaei MR, Rezania SN. An overview on production and application of ferrate (VI) for chemical oxidation, coagulation and disinfection of water and wastewater. J Envir Chem Eng. 2017; 5(2): 1828-1842. https://doi.org/10.1016/j.jece.2017.03.025
[15] Ghernaout D, Naceur MW. Ferrate (VI): In situ generation and water treatment. Desalin Water Treat. 2011;
30(1-3): 1-14. https://doi.org/10.5004/dwt.2011.2217
[16] Munyengabe A, Zvinowanda C. Production, characterization and application of Ferrate(VI) in water and wastewater treatments. Brazilian Journal of Analytical Chemistry. 2019; 6(25): 40-57. https://doi.org/10.30744/brjac.2179-3425.RV-19-2019
[17] Nyzhnyk T. High efficiency titanium coagulants for water treatment. TT: PhE. 2017; 1: 54-56. http://dx.doi.org/10.21303/2585-6847.2017.00486
[18] Zhao YX, Li XY. Polymerized titanium salts for municipal wastewater preliminary treatment followed by further purification via crossflow filtration for water reuse. Sep Purif Technol. 2019; 211: 207-217. https://doi.org/10.1016/j.seppur.2018.09.078
[19] Men Y, Wang X, Cheng S, Zhu L, Li Z. Pre-coagulation with novel titanium coagulants for mitigating membrane fouling in direct membrane filtration of municipal wastewater. Chem Eng J. 2024; 495: 153156. https://doi.org/10.1016/j.cej.2024.153156
[20] Gan Y, Zhang L, Zhang S. The suitability of titanium salts in coagulation removal of micropollutants and in alleviation of membrane fouling. Water Res. 2021; 205: 117692. https://doi.org/10.1016/j.watres.2021.117692
[21] Calderón AJ, González I. Some Hardware and Instrumentation Aspects of the Development of an Automation System for Jar Tests in Drinking Water Treatment. Sensors. 2017; 17: 2305. https://doi.org/10.3390/s17102305
[22] Sarantseva AA, Ivantsova NA, Kuzin EN. Investigation of the Process of Oxidative Degradation of Phenol by Sodium Ferrate Solutions. Russ J Gen Chem. 2023; 93(13): 3454-3459. https://doi.org/10.1134/S1070363223130273
[23] ISO 15705:2002. Water Quality—Determination of the Chemical Oxygen Demand Index (ST‐COD)‐SMALL‐Scale Sealed‐Tube Method; ISO: Geneva, Switzerland. 2002. https://www.iso.org/standard/28778.html
[24] Bain R, Bartram J, Elliott M, Matthews R, McMahan L, Tung R, Chuang P, Gundry S. A summary catalogue of microbial drinking water tests for low and medium resource settings. Int J Environ Res Public Health. 2012; 9: 1609-1625. https://doi.org/10.3390/ijerph9051609
[25] Collivignarelli MC, Abbà A, Benigna I, Sorlini S, Torretta V. Overview of the Main Disinfection Processes for Wastewater and Drinking Water Treatment Plants. Sustainability. 2018; 10(1): 86. https://doi.org/10.3390/su10010086
[26] Kuzin EN, Kruchinina NE. Titanium-containing coagulants. for foundry wastewater treatment. CIS Iron and Steel Review. 2020; 20(2): 66-69. https://doi.org/10.17580/cisisr.2020.02.14
[27] Wang TH, Navarrete‐López AM, Li S, Dixon DA, Gole JL. Hydrolysis of TiCl4: initial steps in the production of TiO2. J Phys Chem A. 2010; 114: 7561-7570. http://dx.doi.org/10.1021/jp102020h
[28] Chekli L, Eripret C, Park S, Tabatabai S, Vronska O, Tamburic B, Kim J, Shon HK. Coagulation performance and floc characteristics of polytitanium tetrachloride (PTC) compared with titanium tetrachloride (TiCl4) and ferric chloride (FeCl3) in algal turbid water. Sep Purif Technol. 2017; 175: 99-106. https://doi.org/10.1016/j.seppur.2016.11.019.
[29] Michael Berg J., Romoser A., Banerjee N. Zebda R., Sayes C. M. The relationship between pH and zeta potential of ∼ 30 nm metal oxide nanoparticle suspensions relevant to in vitro toxicological evaluations. Nanotoxicology. 2009; 3-4: 276-283. https://doi.org/10.3109/17435390903276941